CSPower Lead Carbon Battery – Technology, Advantages
With the progress of society, the requirements for battery energy storage in various social occasions continue to increase. In the past few decades, many battery technologies have made great progress, and the development of lead-acid batteries has also encountered many opportunities and challenges. In this context, scientists and engineers worked together to add carbon to the negative active material of lead-acid batteries, and the lead-carbon battery, an upgraded version of lead-acid batteries, was born.
Lead carbon batteries are an advanced form of Valve Regulated Lead Acid batteries that make use of a cathode made up of carbon and an anode made up of lead. The carbon on the carbon-made cathode performs the function of a capacitor or a ‘supercapacitor’ that allows rapid charging and discharging along with an elongated life at the initial charging stage of the battery.
Why the market need Lead Carbon battery ???
- * Failure modes of flat plate VRLA lead acid batteries in case of intensive cycling
The most common failure modes are:
– Softening or shedding of the active material. During discharge the lead oxide (PbO2) of the positive plate is transformed into lead sulfate (PbSO4), and back to lead oxide during charging. Frequent cycling will reduce cohesion of the positive plate material due to the higher volume of lead sulfate compared to lead oxide.
– Corrosion of the grid of the positive plate. This corrosion reaction accelerates at the end of the charge process due to the, necessary, presence of sulfuric acid.
– Sulfation of the active material of the negative plate. During discharge the lead (Pb) of the negative plate is also transformed into lead sulfate (PbSO4). When left in a low state-of-charge, the lead sulfate crystals on the negative plate grow and harden and form and impenetrable layer that cannot be reconverted into active material. The result is decreasing capacity, until the battery becomes useless.
- * It takes time to recharge a lead acid battery
Ideally, a lead acid battery should be charged a rate not exceeding 0,2C, and the bulk charge phase should be by eight hours of absorption charge. Increasing charge current and charge voltage will shorten recharge time at the expense of reduced service life due to temperature increase and faster corrosion of the positive plate due to the higher charge voltage.
- * Lead carbon: better partial state-of-charge performance, more cycles long life, and higher efficiency deep cycle
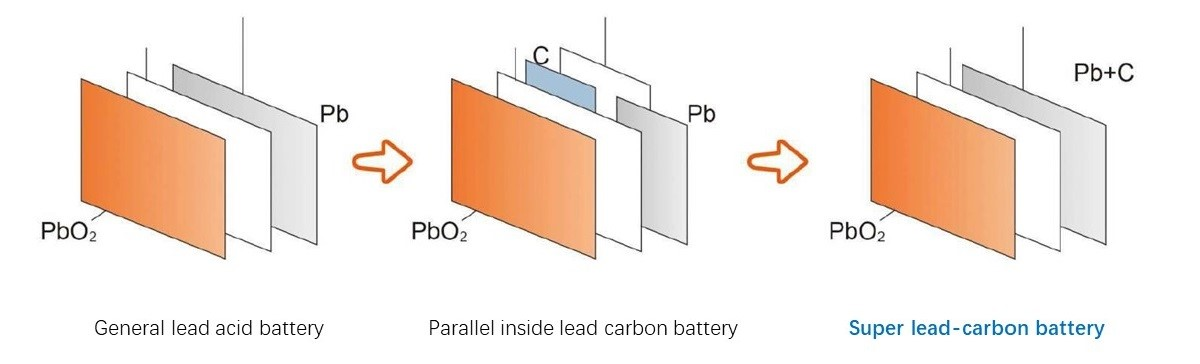
Replacing the active material of the negative plate by a lead carbon composite potentially reduces sulfation and improves charge acceptance of the negative plate.
Lead Carbon Battery Technology
Most of the batteries that are used offer fast charging within an hour or more. While the batteries are under the state of charge, they can still offer output energy which makes them operational even under the state of charge increasing their usage. However, the problem that arose in the lead-acid batteries was that it took a very small time to discharge and a very long time to chargeback again.
The reason that lead-acid batteries took so long to gain their original chargeback was the remnants of lead sulfate that were precipitated on the battery’s electrodes and other internal components. This required an intermittent equalization of the sulfate from electrodes and other battery components. This precipitation of lead sulfate happens with every charge and discharge cycle and the excess of electrons due to precipitation causes hydrogen production resulting in water loss. This problem increases over time and the sulfate remnants begin to form crystals which ruin the charge acceptance capability of the electrode.
The positive electrode of the same battery produces good results despite having the same lead sulfate precipitates which makes it clear that the problem is within the negative electrode of the battery. To overcome this issue, scientists and manufacturers have solved this problem by adding carbon to the negative electrode (cathode) of the battery. The addition of carbon improves the battery’s charge acceptance eliminating the partial charge and aging of the battery due to lead sulfate remnants. By adding carbon, the battery starts to behave as a ‘supercapacitor’ offering its properties for better performance of the battery.
The lead-carbon batteries are a perfect replacement for applications that involve a lead-acid battery like in frequent start-stop applications and micro/mild hybrid systems. Lead-carbon batteries can be heavier as compared to other types of batteries but they are cost-effective, resistant to extreme temperatures, and do not require cooling mechanisms to work alongside them. In contrary to the traditional lead-acid batteries, these lead-carbon batteries operate perfectly between 30 and 70 percent charging capacity without the fear of sulfate precipitations. Lead-carbon batteries have outperformed the lead-acid batteries in most of the functions but they suffer a voltage drop on discharge as a supercapacitor does.
Construction for CSPower Fast Charge Deep Cycle Lead Carbon battery
Features for Fast Charge Deep Cycle Lead Carbon battery
- l Combine the characteristics of lead acid battery and super capacitor
- l Long life cycle service design, excellent PSoC and cyclic performance
- l High power, rapid charging and discharging
- l Unique grid and lead pasting design
- l Extreme temperature tolerance
- l Able to operate at -30°C -60°C
- l Deep Discharge recovery capability
Advantages for Fast Charge Deep Cycle Lead Carbon battery
Every battery has its designated use depending on its applications and can not be termed as good or bad in a general way.
A lead-carbon battery may not be the most recent technology for batteries but it does offer some great advantages that even the recent battery technologies can not offer. Some of these advantages of lead-carbon batteries are given below:
- l Less sulfation in case of partial state-of-charge operation.
- l Lower charge voltage and therefore higher efficiency and less corrosion of the positive plate.
- l And the overall result is improved cycle life.
Tests have shown that our lead carbon batteries do withstand at least eight hundred 100% DoD cycles.
The tests consist of a daily discharge to 10,8V with I = 0,2C₂₀, by approximately two hours rest in discharged condition, and then a recharge with I = 0,2C₂₀.
- l ≥ 1200 cycles @ 90% DoD (discharge to 10,8V with I = 0,2C₂₀, by approximately two hours rest in discharged condition, and then a recharge with I = 0,2C₂₀)
- l ≥ 2500 cycles @ 60% DoD (discharge during three hours with I = 0,2C₂₀, immediately by recharge at I = 0,2C₂₀)
- l ≥ 3700 cycles @ 40% DoD (discharge during two hours with I = 0,2C₂₀, immediately by recharge at I = 0,2C₂₀)
- l The thermal damage effect is minimal in lead-carbon batteries due to their charge-discharge properties. Individual cells are far from the risks of burning, exploding, or overheating.
- l Lead-carbon batteries are a perfect match for on-grid and off-grid systems. This quality makes them a good choice for solar electricity systems because they offer high discharge current ability
Lead carbon batteries VS Sealed lead acid battery , gel batteries
- l Lead carbon batteries are better at sitting at partial states of charge (PSOC). Ordinary lead type batteries work best and last longer if they follow a strict ‘full charge’-‘full discharge’-full charge’ regime; they do not respond well to being charged at any state in between full and empty. Lead carbon batteries are happier to function in the more ambiguous charging regions.
- l Lead Carbon batteries use supercapacitor negative electrodes. Carbon batteries use a standard lead type battery positive electrode and a supercapacitor negative electrode. This supercapacitor electrode is the key to the longevity of the carbon batteries. A standard lead-type electrode undergoes a chemical reaction over time from charging and discharging. The supercapacitor negative electrode reduces corrosion on the positive electrode and that leads to longer life of the electrode itself which then leads to longer-lasting batteries.
- l Lead carbon batteries have faster charge/discharge rates. Standard lead-type batteries have between maximum 5-20% of their rated capacity charge/discharge rates meaning you can charge or discharge the batteries between 5 – 20 hours without causing any long term damage to the units. Carbon Lead has a theoretical unlimited charge/discharge rate.
- l Lead carbon batteries do not require any maintenance. The batteries are fully sealed and don’t require any active maintenance.
- l Lead carbon batteries are cost-competitive with gel type batteries. Gel batteries are still slightly cheaper to buy upfront, but carbon batteries are only slightly more. The current price difference between Gel and Carbon batteries is roughly 10-11%. Take into consideration that carbon lasts for approximately 30% longer and you can see why it’s a better value for money option.
Post time: Apr-08-2022